Hydrazine: Difference between revisions
More actions
Footgunning myself at this point |
Fixed incorrect description stating Hydrazine was hypergolic & added some links. |
||
| (5 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
{{Infobox gas | {{Infobox gas | ||
| gas_name = Hydrazine | | gas_name = Hydrazine | ||
| image = Icon- | | image = Icon-Hydrazine.png | ||
| heat = 48.4 J/mol*K <!-- Chemistry.SPECIFIC_HEAT_HYDRAZINE --> | | heat = 48.4 J/mol*K <!-- Chemistry.SPECIFIC_HEAT_HYDRAZINE --> | ||
| latent_heat = 4000 J/mol <!-- LATENT_HEAT_HYDRAZINE --> | | latent_heat = 4000 J/mol <!-- LATENT_HEAT_HYDRAZINE --> | ||
| Line 11: | Line 11: | ||
| coefficient_b = 9.15642808045339 <!-- Chemistry.COEFFIENT_B_HYDRAZINE (sic) --> | | coefficient_b = 9.15642808045339 <!-- Chemistry.COEFFIENT_B_HYDRAZINE (sic) --> | ||
| thermal_efficiency = 0.08 <!-- Liquid hydrazine: 0.00 --> | | thermal_efficiency = 0.08 <!-- Liquid hydrazine: 0.00 --> | ||
| enthalpy = | | enthalpy = 306 kJ / mol <!-- Chemistry.ENTHALPY_HYDRAZINE --> | ||
| liquid_volume = 30 L/kmol <!--Chemistry.MOLAR_VOLUME_HYDRAZINE--> | | liquid_volume = 30 L/kmol <!--Chemistry.MOLAR_VOLUME_HYDRAZINE--> | ||
| molar_mass = 32 g/mol | | molar_mass = 32 g/mol | ||
| heat_capacity_ratio = 1.26 <!--Chemistry.PolyatomicDegreesOfFreedom--> | | heat_capacity_ratio = 1.26 <!--Chemistry.PolyatomicDegreesOfFreedom--> | ||
}} | }} | ||
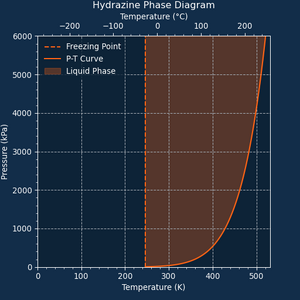
[[File:Phase Diagram of Hydrazine.png|thumb|Phase Diagram of Hydrazine]] | |||
== Description == | == Description == | ||
<blockquote><q>Hydrazine is the perfect fuel for space exploration as it requires no oxidiser to combust, only needing heat or a spark. It is more commonly stored in its liquid form Liquid Hydrazine which is stable at room temperature. Hydrazine is extremely reactive and will violently combust in the presence of an oxidiser. It is extremely toxic to all forms of life.</q><br> | <blockquote><q>Hydrazine is the perfect fuel for space exploration as it requires no oxidiser to combust, only needing heat or a spark. It is more commonly stored in its liquid form Liquid Hydrazine which is stable at [[room]] temperature. Hydrazine is extremely reactive and will violently combust in the presence of an oxidiser. It is extremely toxic to all forms of life.</q><br> | ||
'''- Stationpedia'''</blockquote> | '''- [[Stationpedia]]'''</blockquote> | ||
'''Hydrazine''' or '''N<sub>2</sub>H<sub>4</sub>''' is a [[ | '''Hydrazine''' or '''N<sub>2</sub>H<sub>4</sub>''' is a [[wikipedia:Monopropellant|Monopropellant]] that ignites at 520.8 K (247.7°C), or when sparked. | ||
=== Combustion === | === Combustion === | ||
* 1 | * {{Combustion|Fuel=Hydrazine, 1|OutputA=Pollutant, 4|ShowAutoignition=true}} | ||
== Usage == | == Usage == | ||
''See also: [[Fuel]]'' | ''See also: [[Fuel]]'' | ||
Hydrazine has a low combustion enthalpy, making it suboptimal for rocketry applications that require high performance. However, | Hydrazine has a low [[wikipedia:Heat_of_combustion|combustion enthalpy]], making it suboptimal for [[rocketry]] applications that require high performance. However, being a monopropellant allows its use in a [[Pumped Gas Engine]] or [[Pressure-Fed Liquid Engine]] without requiring any premixing, simplifying rocket and/or launch pad design. | ||
===Notes=== | ===Notes=== | ||
* Hydrazine is extremely toxic to all forms of life, causing damage in concentrations above 0.1% for [[plants]], or 1kPa for Humans and Zrilians. The increased toxicity to plants is only shared with [[Hydrochloric Acid]]. | |||
==Sources== | ==Sources== | ||
Hydrazine can be acquired from [[traders]]. | |||
{| class="wikitable" | {| class="wikitable" | ||
|- | |- | ||
! Source !! Amount | ! Source !! Amount | ||
|- | |- | ||
| ? || ? | |? || ? | ||
|} | |} | ||
Latest revision as of 05:08, 3 April 2026
Description
Hydrazine is the perfect fuel for space exploration as it requires no oxidiser to combust, only needing heat or a spark. It is more commonly stored in its liquid form Liquid Hydrazine which is stable at room temperature. Hydrazine is extremely reactive and will violently combust in the presence of an oxidiser. It is extremely toxic to all forms of life.
- Stationpedia
Hydrazine or N2H4 is a Monopropellant that ignites at 520.8 K (247.7°C), or when sparked.
Combustion
Usage
See also: Fuel
Hydrazine has a low combustion enthalpy, making it suboptimal for rocketry applications that require high performance. However, being a monopropellant allows its use in a Pumped Gas Engine or Pressure-Fed Liquid Engine without requiring any premixing, simplifying rocket and/or launch pad design.
Notes
- Hydrazine is extremely toxic to all forms of life, causing damage in concentrations above 0.1% for plants, or 1kPa for Humans and Zrilians. The increased toxicity to plants is only shared with Hydrochloric Acid.
Sources
Hydrazine can be acquired from traders.
| Source | Amount |
|---|---|
| ? | ? |
References
Gases and Liquids
| |||
|---|---|---|---|
| Elements | Compounds | Mixtures | Other |
|
|
|||
